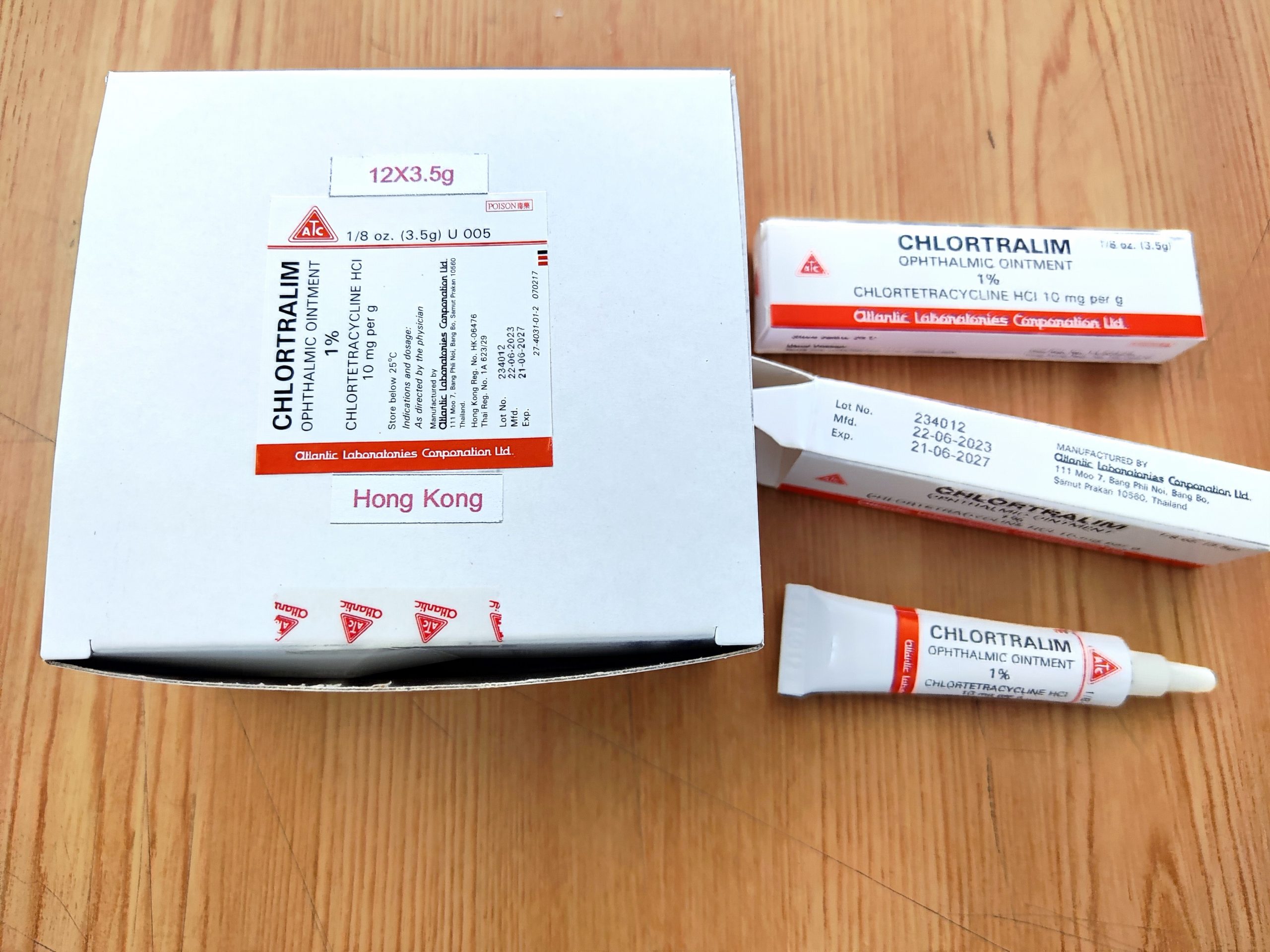
The Department of Health (DH) endorsed Atlantic Pharmaceutical Ltd (Atlantic) on 29 November 2024, to recall the Batch No. 234012 from the market as a precautionary measure due to reported presence of sediments publishes information by The Health Science Authority (HSA) in Singapore.
The above affected batch of the product have been imported into Hong Kong and supplied to HA 3 hospitals, 2 local doctors, and pharmacies.
Atlantic Pharmaceutical Ltd (Atlantic) has set up a dedicated Recall hotline
(852 – 2563 4224) to handle related enquiries.
Atlantic HK will closely monitor the recall and report to DH as appropriate.
So far, Atlantic HK has not received any adverse reaction reports in connection with the impacted batches.
Monday December 2, 2024